Understanding Mitochondria:The Foundation of Cellular Energy
How these dynamic organelles power adaptation, recovery, and long-term health.
Table of Contents
What are Mitochondria?
Mitochondria are membrane-bound organelles found in nearly all human cells. While they perform various essential functions, such as regulating calcium homeostasis, producing reactive oxygen species for signaling, contributing to biosynthesis of macromolecules, influencing cell death (apoptosis), and maintaining redox balance, their most well-known and perhaps most important role is serving as the primary site for producing energy (ATP) through aerobic respiration (oxidative phosphorylation).
They are especially abundant in energy-demanding cells like muscle cells (hundreds to thousands per cell), liver cells, neurons, and kidney cells.
Mitochondria are unique among cellular structures because they contain their own DNA, separate from the DNA found in the cell nucleus.
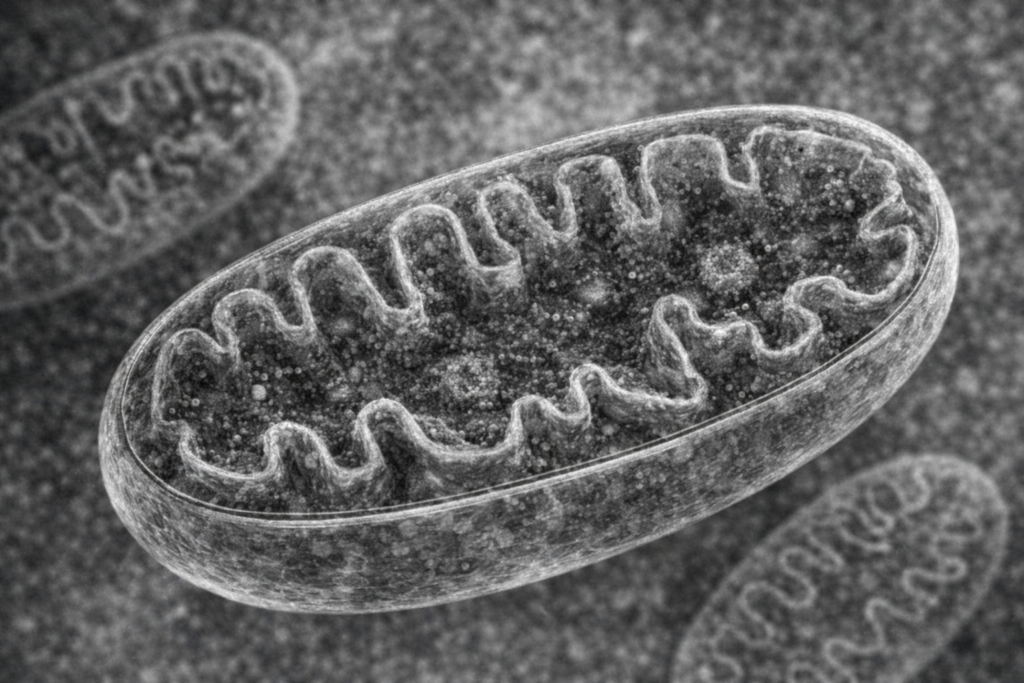
A Brief History of Mitochondria
How we came to have mitochondria in our bodies and why they have their own DNA is very interesting scientifically!
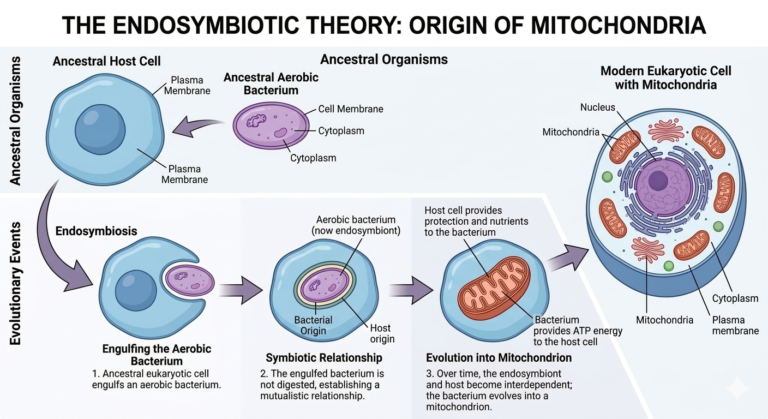
The most widely accepted explanation is called the endosymbiotic theory.
According to this theory, mitochonria likely became part of our cells over a billion years ago, when a larger ancestral single-celled organism engulfed a smaller aerobic bacterium capable of efficient energy production. Instead of being digested, the bacterium survived inside the host cell.
Over time, the two formed a stable, mutually beneficial relationship and co-evolved. The bacterium gradually became what we now call the mitochondrion.
This evolutionary origin helps explain why mitochondria still contain their own small circular DNA and why they remain essential for cellular energy production today.
How much mitochondria do we have?
The number of mitochondria within a cell is closely linked to its energy requirements. Tissues that sustain continuous or metabolically intensive activity, such as heart, liver, and muscle, contain large numbers of mitochondria, while mature red blood cells contain none. The approximate values below illustrate how cellular energy demand influences mitochondrial abundance across human tissues.
| Cell type | Approx. mitochondria per cell | Notes |
|---|---|---|
| Red blood cells (erythrocytes) | 0 | Mature erythrocytes lack mitochondria to maximize hemoglobin content and oxygen transport capacity. |
| White blood cells (leukocytes) | 3 – 100 | Typically low at rest (e.g., lymphocytes); higher in more metabolically active/activated immune cells. |
| Epithelial cells | 100 – 500 | Moderate abundance; varies by tissue and transport workload (e.g., renal tubular epithelium is higher). |
| Neurons | 100 – 2,000 | Variable; enriched in regions with high ATP demand (synapses, nodes, and long axons). |
| Liver cells (hepatocytes) | 800 – 2,000 | High abundance reflecting intensive metabolic flux and detoxification pathways. |
| Skeletal muscle fibers | 200 – 5,000+ | Highly variable; depends on fiber type and training status. Endurance training increases mitochondrial density. |
| Cardiac muscle cells (cardiomyocytes) | 3,000 – 8,000 | Very high abundance to sustain continuous contractile activity. |
| Oocytes (egg cells) | 100,000 – 600,000+ | Extremely high abundance to support early embryonic development prior to robust mitochondrial biogenesis. |
| Other somatic cells (typical range) | 100 – 1,000 | Common range for many non-specialized cell types (e.g., fibroblasts). |
Values are approximate and vary by tissue type, physiological state, species, and measurement method.
What do mitochondria do?
Mitochondria are tiny structures inside your cells that convert nutrients and oxygen into ATP, the main “spendable” energy your cells use to function. Beyond energy supply, they also help coordinate metabolism, stress responses, and cellular maintenance.
Key takeaway
Mitochondria are best understood as energy converters and control hubs that help cells adapt to demand.
Fuel + oxygen → electron transport → ATP + heat
Make usable cellular energy (ATP)
Through aerobic metabolism, mitochondria produce most ATP in many cell types—fueling contraction, transport, repair, and signaling.
Choose and process fuel
They help the cell “decide” how to use fats, carbohydrates, and other substrates depending on availability and demand.
Coordinate cellular signals
They generate signaling molecules (including reactive oxygen species in controlled amounts) that influence adaptation and gene expression.
Manage stress, calcium, and cell fate
They buffer calcium and participate in quality-control pathways; under severe stress they can also help initiate programmed cell death (apoptosis).
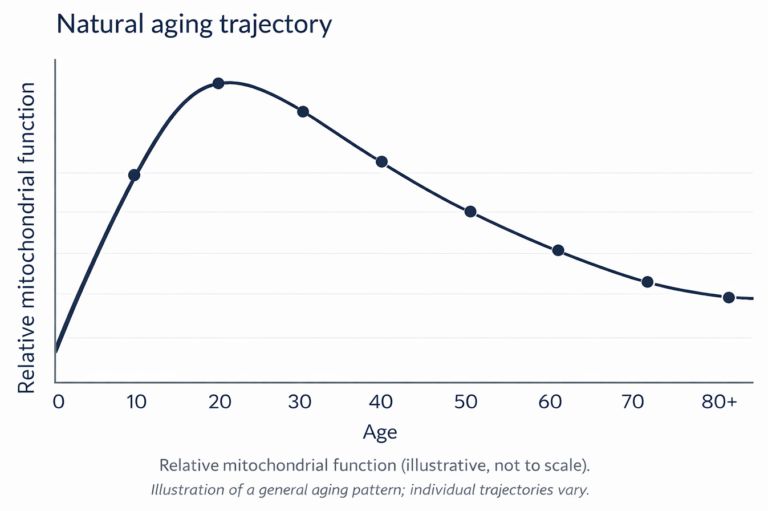
Mitochondrial Decline: Aging + Lifestyle Factors
Mitochondrial function tends to peak in early adulthood, then gradually declines with age, at roughly 10–15% per decade.
Beyond aging, lifestyle can accelerate that decline:
Poor diet
more metabolic strain, fewer nutrients
Poor sleep
less cellular repair and recovery, more stress on mitochondria
Lack of exercise
fewer signals to maintain capacity (use it or lose it)
Chronic stress
sustained stress signaling can disrupt energy regulation and increase inflammatory/oxidative burden
What happens when mitochondria don’t function properly?
When mitochondria underperform, cells may:
- Produce less ATP, the energy required for normal cellular function
- Have less capacity to support everyday demands
- Become less efficient at using fuel
- Generate more stress signals, including oxidative stress
- Shift resources toward essential tasks and away from repair and recovery
Key takeaway
Mitochondrial dysfunction can reduce cellular energy availability and increase stress signaling, two factors that can affect how well the body adapts and recovers.
Why it matters: Energy isn’t just how you feel. It’s what cells can do.
Faster fatigue
Cells have less “spendable” energy for high-demand tasks like contraction, transport, and repair.
Altered fuel handling
Cells may rely more on less efficient pathways or struggle to switch cleanly between fats and carbohydrates.
Slower covery
After workouts, poor sleep, or busy weeks, it fan take longer to feel back to baseline.
Lower exercise tolerance
Activities that used to feel easy may feel harder at the same pace or intensity.
(These are common experiences with many possible causes. This section is educational, not diagnostic.)
What influences mitochondrial function
Sleep quality and circadian regularity
Physical activity
Nutrition and total energy intake
Chronic stress load
Environmental exposures
Age, medications, underlying context
Educational information only, not medical advice. If symptoms are persistent or severe, consult a qualified clinician.
How can mitochondrial function be supported?
Mitochondria are dynamic structures that respond to demand. Their function is influenced by movement, sleep, nutrition, stress exposure, and overall metabolic health.
Mitochondrial support is best handled through consistent lifestyle inputs that signal adaptation, repair, and renewal at the cellular level.
Key takeaway
Mitochondrial health is shaped by daily signals, movement, recovery, and metabolic balance, not by isolated interventions.
Energy capacity reflects how well cells adapt over time.
Regular Aerobic & Resistance Training
Physical activity stimulates mitochondrial biogenesis and improves efficiency of energy production pathways.
Sleep & Circadian Regularity
Deep, consistent sleep supports cellular repair, antioxidant balance, and metabolic regulation.
Nutritional Quality & Energy Balance
Adequate protein, micronutrients, and total energy intake support mitochondrial structure and function.
Stress Load & Recovery Cycles
Chronic stress without recovery can strain cellular energy systems. Periods of rest help restore balance.
Environmental & Metabolic Context
Tobacco smoke, excess alcohol, and pollutants can strain mitochondria and increase oxidative stress.
Supplementation
Consider Mitozz if you want focused support for mitochondrial function and sustained energy at the cellular level.
Not sure which mitochondrial supplements are actually worth looking at?
Many products are marketed for energy, aging, and cellular health, but their mechanisms are not the same.
Explore our comparison page to see how Mitozz stacks up against other well-known mitochondrial supplements.
Want a deeper, science-based breakdown?
We explore the mechanisms, lifestyle strategies, and emerging research in detail in our guide:
How to Repair and Maintain Mitochondrial Health Naturally
