Mitozz Testimonial Collage

What do people actually notice when they start Mitozz? They talk about steadier energy, clearer focus, easier movement, and a calmer body feel. This testimonial collage highlights the most common “first changes” people report with Mitozz and explains the physiology behind them.
Can Epicatechin Help Long COVID Fatigue? New Clinical Trial on Inflammation and Cellular Energy
What biological mechanisms drive persistent long COVID fatigue? A new placebo-controlled clinical trial examined this question and tested whether epicatechin could influence them. Here’s what the data showed.
Dark Chocolate, Epicatechin, and Cellular Energy

Chocolate is often enjoyed for its flavor, but research into cocoa points to something deeper. Discover how epicatechin has been studied for its role in supporting how cells produce and manage energy.
(−)-Epicatechin reduces muscle waste after complete spinal cord transection in a murine model: role of ubiquitin–proteasome system
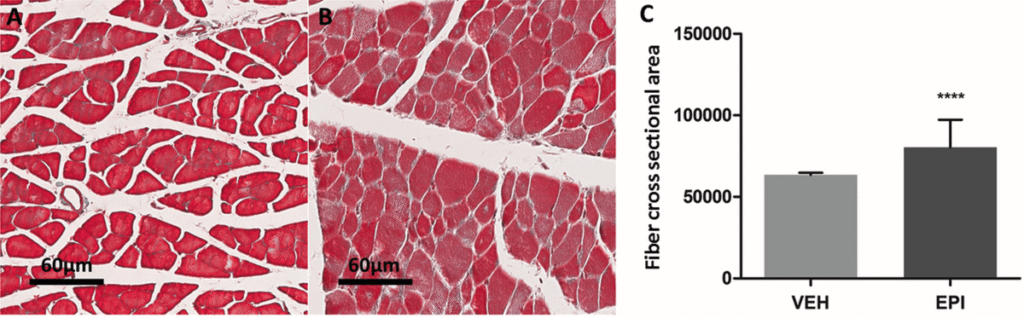
The skeletal muscle mass reduces 30–60% after spinal cord injury, this is mostly due to protein degradation through ubiquitin–proteasome system. In this work, we propose that the flavanol (−)-epicatechin, due its widespread biological effects on muscle health, can prevent muscle mass decrease after spinal cord injury. Thirty-six female Long Evans rats were randomized into 5 groups: (1) Spinal cord injury 7 days, (2) Spinal cord injury + (−)-epicatechin 7 days, (3) Spinal cord injury 30 days, (4) Spinal cord injury + (−)-epicatechin 30 days and (5) Sham (Only laminectomy). Hind limb perimeter, muscle cross section area, fiber cross section area and ubiquitin–proteasome system protein expression together with total protein ubiquitination were assessed. At 30 days Spinal cord injury group lost 49.52 ± 2.023% of muscle cross section area (−)-epicatechin treated group lost only 24.28 ± 15.45% being a significant difference. Ubiquitin–proteasome markers showed significant changes. FOXO1a increased in spinal cord injury group vs Sham (−)-epicatechin reduced this increase. In spinal cord injury group MAFbx increased significantly vs Sham but decrease in (−)-epicatechin treatment group at 30 days. At 7 and 30 days MuRF1 increased in the spinal cord injury and decreased in the (−)-epicatechin group. The global protein ubiquitination increases after spinal cord injury, epicatechin treatment induce a significant decrease in protein ubiquitination. These results suggest that (−)-epicatechin reduces the muscle waste after spinal cord injury through down regulation of the ubiquitin–proteasome system.
Anticancer potential of (−)-epicatechin in atriple-negative mammary gland model
Objectives The main aim of this work was to analyse the potential tumour growth inhibition effects
of (−)-epicatechin (EC). Triple-negative breast cancer (TNBC) is an invasive form of cancer charac-
terized by the absence of progesterone receptor, estrogen receptor and human epidermal growth
factor receptor 2. Doxorubicin (DOX) is widely used for its anti-tumour activity. EC belongs to the
flavanol subfamily and is a candidate molecule for the adjuvant treatment of cancer due to its
antiproliferative activities.
Methods Evaluation of EC effects and pathways involved in a model of TNBC.
Key findings EC inhibited tumour growth as efficiently as DOX (inhibition rates of 74% and 79% for
EC and DOX, respectively). The evaluation of adenosine monophosphate-activated protein kinase
(AMPK) and Akt phosphorylation and mTOR expression indicates that EC modulates these path-
ways, resulting in the inhibition of cell proliferation. Additionally, we found an increase in the sur-
vival of EC-treated animals compared with control-treated animals. This effect was similar to the
effects induced by DOX (survival rates of 44% and 30% for EC and DOX, respectively).
Conclusion EC has antiproliferative properties and increases survival in a model of TNBC. These ef-
fects may occur through the modulation of deregulated AMPK and Akt/mTOR signalling pathways.
