Mitochondrial Biogenesis, Muscle Regeneration, and Human Neuromuscular Disease

In adults with Becker muscular dystrophy, (-)-epicatechin increased markers of mitochondrial biogenesis and muscle regeneration, suggesting a potentially relevant biological response in a human neuromuscular condition.
Beneficial Effects of Flavonoids on Skeletal Muscle Health: A Systematic Review and Meta-Analysis
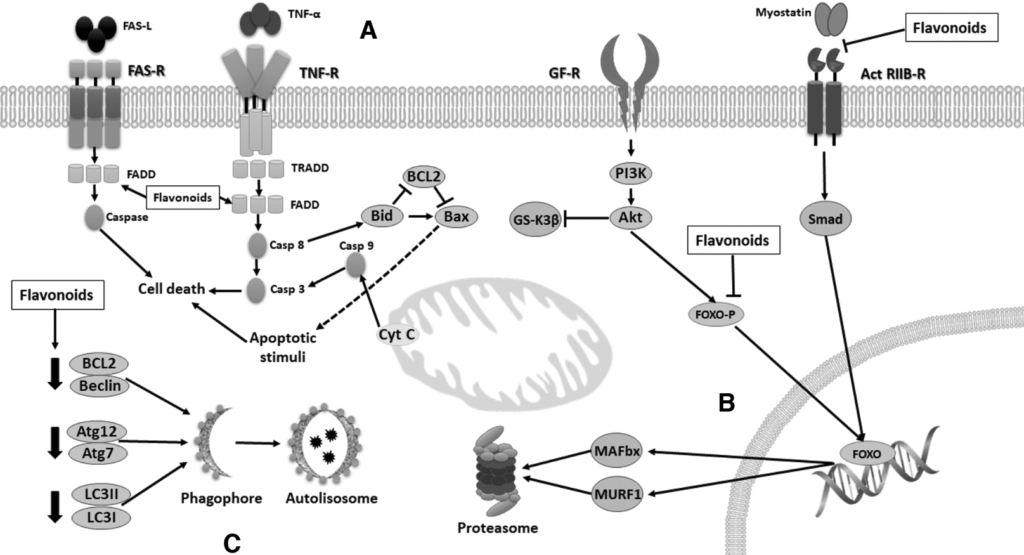
Skeletal muscle (SkM) is a highly dynamic tissue that responds to physiological adaptations or pathological conditions, and SkM mitochondria play a major role in bioenergetics, regulation of intracellular calcium homeostasis, pro-oxidant/antioxidant balance, and apoptosis. Flavonoids are polyphenolic compounds with the ability to modulate molecular pathways implicated in the development of mitochondrial myopathy. Therefore, it is pertinent to explore its potential application in conditions such as aging, disuse, denervation, diabetes, obesity, and cancer. To evaluate preclinical and clinical effects of flavonoids on SkM structure and function. We performed a systematic review of published studies, with no date restrictions applied, using PubMed and Scopus. The following search terms were used: “flavonoids” OR “flavanols” OR “flavones” OR “anthocyanidins” OR “flavanones” OR “flavan-3-ols” OR “catechins” OR “epicatechin” OR “(−)-epicatechin” AND “skeletal muscle.” The studies included in this review were preclinical studies, clinical trials, controlled clinical trials, and randomized-controlled trials that investigated the influence of flavonoids on SkM health. Three authors, independently, assessed trials for the review. Any disagreement was resolved by consensus. The use of flavonoids could be a potential tool for the prevention of muscle loss. Their effects on metabolism and on mitochondria function suggest their use as muscle regulators.
(−)-Epicatechin reduces muscle waste after complete spinal cord transection in a murine model: role of ubiquitin–proteasome system
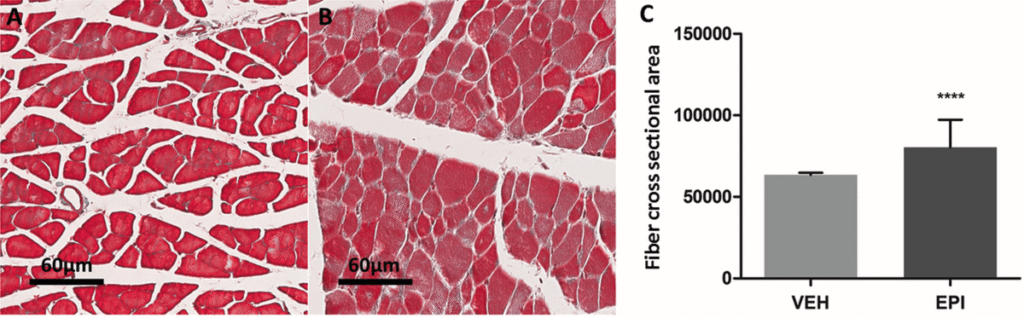
The skeletal muscle mass reduces 30–60% after spinal cord injury, this is mostly due to protein degradation through ubiquitin–proteasome system. In this work, we propose that the flavanol (−)-epicatechin, due its widespread biological effects on muscle health, can prevent muscle mass decrease after spinal cord injury. Thirty-six female Long Evans rats were randomized into 5 groups: (1) Spinal cord injury 7 days, (2) Spinal cord injury + (−)-epicatechin 7 days, (3) Spinal cord injury 30 days, (4) Spinal cord injury + (−)-epicatechin 30 days and (5) Sham (Only laminectomy). Hind limb perimeter, muscle cross section area, fiber cross section area and ubiquitin–proteasome system protein expression together with total protein ubiquitination were assessed. At 30 days Spinal cord injury group lost 49.52 ± 2.023% of muscle cross section area (−)-epicatechin treated group lost only 24.28 ± 15.45% being a significant difference. Ubiquitin–proteasome markers showed significant changes. FOXO1a increased in spinal cord injury group vs Sham (−)-epicatechin reduced this increase. In spinal cord injury group MAFbx increased significantly vs Sham but decrease in (−)-epicatechin treatment group at 30 days. At 7 and 30 days MuRF1 increased in the spinal cord injury and decreased in the (−)-epicatechin group. The global protein ubiquitination increases after spinal cord injury, epicatechin treatment induce a significant decrease in protein ubiquitination. These results suggest that (−)-epicatechin reduces the muscle waste after spinal cord injury through down regulation of the ubiquitin–proteasome system.
(-)-Epicatechin improves mitochondrial-related protein levels and ameliorates oxidative stress in dystrophic δ-sarcoglycan null mouse striated muscle
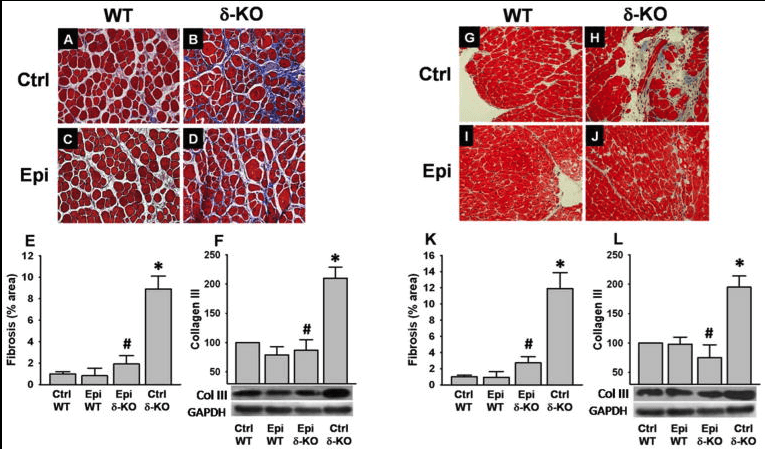
Muscular dystrophies (MDs) are a group of heterogeneous genetic disorders characterized by progressive striated muscle wasting and degeneration. Although the genetic basis for many of these disorders has been identified, the exact mechanism of disease pathogenesis remains unclear. The presence of oxidative stress (OS) is known to contribute to the pathophysiology and severity of the MD. Mitochondrial dysfunction is observed in MD, and probably represents an important determinant of increased OS. Experimental antioxidant therapies have been implemented with the aim of protecting against disease progression, but results from clinical trials have been disappointing. In this study, we explored the capacity of the cacao flavonoid (-)-epicatechin (Epi) to mitigate OS by acting as a positive regulator of mitochondrial structure/function endpoints and redox balance control systems in skeletal and cardiac muscles of dystrophic, δ-sarcoglycan (δ-SG) null mice. Wild-type or δ-SG null 2.5-month-old male mice were treated via oral gavage with either water (controls) or Epi (1 mg·kg(-1) , twice daily) for 2 weeks. The results showed significant normalization of total protein carbonylation, recovery of the glutathione/oxidized glutathione ratio and enhanced superoxide dismutase 2, catalase and citrate synthase activities with Epi treatment. These effects were accompanied by increases in the protein levels of thioredoxin, glutathione peroxidase, superoxide dismutase 2, catalase, and mitochondrial endpoints. Furthermore, we found decreases in heart and skeletal muscle fibrosis, accompanied by an improvement in skeletal muscle function, with treatment. These results warrant further investigation of Epi as a potential therapeutic agent to mitigate MD-associated muscle degeneration.
PXR is a target of (-)-epicatechin in skeletal muscle
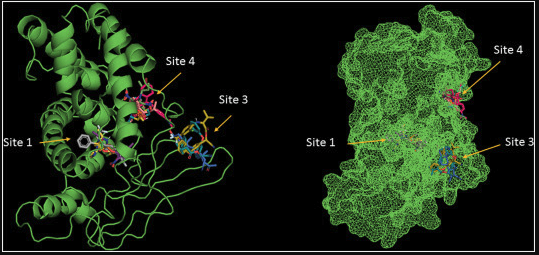
(-)-Epicatechin (EC) is a flavanol that has shown numerous biological effects such as: decrease risk of cardiovascular dysfunction, metabolism regulation, skeletal muscle (SkM) performance improvement and SkM cells differentiation induction, among others. The described EC acceptor/receptor molecules do not explain the EC’s effect on SkM. We hypothesize that the pregnane X receptor (PXR) can fulfill those characteristics, based on structural similitude between EC and steroidal backbone and that PXR activation leads to similar effects as those induced by EC. In order to demonstrate our hypothesis, we: 1) analyzed the possible EC and mouse PXR interaction through in silico strategies, 2) developed an EC’s affinity column to isolate PXR, 3) evaluated, in mouse myoblast (C2C12 cells) the inhibition of EC-induced PXR’s nucleus translocation by ketoconazole, a specific blocker of PXR and 4) analyzed the effect of EC as an activator of mouse PXR, evaluating the expression modulation of cytochrome 3a11 (Cyp3a11) gen and myogenin protein. (-)-Epicatechin interacts and activates PXR, promoting this protein translocation to the nucleus, increasing the expression of Cyp3a11, and promoting C2C12 cell differentiation through increasing myogenin expression. These results can be the base of further studies to analyze the possible participation of PXR in the skeletal muscle effects shown by EC.
Brain Protein Structure and Cellular Stability

This study evaluated whether (-)-epicatechin could restore dystrophin-associated protein complex (DAPC) components and dysbindin in the prefrontal cortex of mdx mice, a model of Duchenne muscular dystrophy. After four weeks of treatment, (-)-epicatechin partially recovered multiple DAPC-related proteins and associated interactions, suggesting a shift toward a healthier brain protein profile.
(-)-Epicatechin Inhibits Metastatic-Associated Proliferation, Migration, and Invasion of Murine Breast Cancer Cells In Vitro
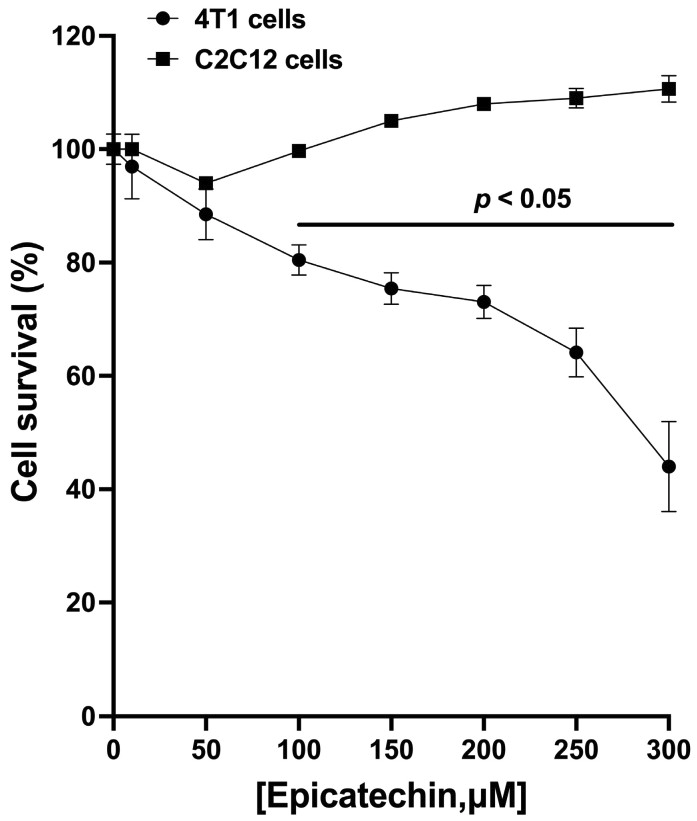
Breast cancer, due to its high incidence and mortality, is a public health problem worldwide. Current chemotherapy uses non-specific cytotoxic drugs, which inhibit tumor growth but cause significant adverse effects. (-)-Epicatechin (EC) is part of a large family of biomolecules called flavonoids. It is widely distributed in the plant kingdom; it can be found in green tea, grapes, and cocoa. Several studies in animals and humans have shown that EC induces beneficial effects in the skeletal muscle and the cardiovascular system, reducing risk factors such as arterial hypertension, endothelial dysfunction, damage to skeletal muscle structure, and mitochondrial malfunction by promoting mitochondrial biogenesis, with no adverse effects reported. Recently, we reported that EC had an antitumor effect in a murine triple-negative mammary gland tumor model, decreasing tumoral size and volume and increasing survival by 44%. This work aimed to characterize the effects of flavanol EC on proliferation, migration, and metastasis markers of triple-negative murine breast (4T1) cancer cells in culture. We found proliferation diminished and Bax/Bcl2 ratio increased. When the migration of culture cells was evaluated, we observed a significant reduction in migration. Also, the relative expression of the genes associated with metastasis, Cdh1, Mtss1, Pten, Bmrs, Fat1, and Smad4, was increased. In conclusion, these results contribute to understanding molecular mechanisms activated by EC that can inhibit metastatic-associated proliferation, migration, and invasion of murine breast cancer cells.
CHARACTERIZATION OF THE CYTOTOXIC EFFECTSOF THE COMBINATION OF CISPLATIN AND FLAVANOL(-)-EPICATECHIN ON HUMAN LUNG CANCER CELL LINE A549. AN ISOBOLOGRAPHIC APPROACH
Among malignancies, lung cancer is a leading cause of death. Platinum-based therapeutic compounds used to treat lung cancer have not been able to increase the survival of patients and such compounds have a high incidence of adverse and toxic effects. It has been proposed that flavonoids such as catechins may significantly reduce the risk of developing cancer, alongside with other health benefits. The aim of this work was to determine the effect of (-)-epicatechin, the main flavanol found in cocoa, on the proliferation of the lung non-small cell adenocarcinoma cancer cell line A549, and to determine its effects when added si- multaneously with cisplatin. Materials and Methods: Concentration-response curves for cisplatin and epicatechin were obtained, inhibitory concentrations calculated and an isobolographic analysis was then performed.
Anticancer potential of (−)-epicatechin in atriple-negative mammary gland model
Objectives The main aim of this work was to analyse the potential tumour growth inhibition effects
of (−)-epicatechin (EC). Triple-negative breast cancer (TNBC) is an invasive form of cancer charac-
terized by the absence of progesterone receptor, estrogen receptor and human epidermal growth
factor receptor 2. Doxorubicin (DOX) is widely used for its anti-tumour activity. EC belongs to the
flavanol subfamily and is a candidate molecule for the adjuvant treatment of cancer due to its
antiproliferative activities.
Methods Evaluation of EC effects and pathways involved in a model of TNBC.
Key findings EC inhibited tumour growth as efficiently as DOX (inhibition rates of 74% and 79% for
EC and DOX, respectively). The evaluation of adenosine monophosphate-activated protein kinase
(AMPK) and Akt phosphorylation and mTOR expression indicates that EC modulates these path-
ways, resulting in the inhibition of cell proliferation. Additionally, we found an increase in the sur-
vival of EC-treated animals compared with control-treated animals. This effect was similar to the
effects induced by DOX (survival rates of 44% and 30% for EC and DOX, respectively).
Conclusion EC has antiproliferative properties and increases survival in a model of TNBC. These ef-
fects may occur through the modulation of deregulated AMPK and Akt/mTOR signalling pathways.
