Your cells are in constant communication. Every time your immune system detects a threat, it sends signals that ripple through your biology, down to the level of individual organelles. And your mitochondria, the structures responsible for producing most of your cellular energy, are far more involved in that signaling than most people realize. Understanding the relationship between mitochondria and chronic inflammation is one of the more important insights to emerge from cell biology in recent decades, because it helps explain why inflammation that lingers can affect how your body feels and functions over time.
Why Mitochondria Are Central to Immune Signaling
Mitochondria have long been described primarily as energy producers. That’s accurate but incomplete. These organelles also play a significant role in regulating the immune response. They sit at an intersection of cellular metabolism, redox signaling, and innate immunity, meaning they help determine not only how much energy a cell has, but how that cell responds to stress and threats.
Research over the past two decades has shown that mitochondria act as a kind of platform for inflammatory signaling. When they are functioning well, they help calibrate immune responses, supporting activation when needed and resolution when the threat has passed. When they are not functioning well, that calibration can shift, and inflammation can become more persistent.
How Dysfunctional Mitochondria Fuel Inflammation
The clearest mechanism here involves reactive oxygen species (ROS), molecules produced as a natural byproduct of energy metabolism. Under normal conditions, ROS are produced in small amounts and neutralized efficiently by the cell’s antioxidant systems. Under conditions of cellular stress, however, mitochondria can produce ROS in excess, and that excess is where the relationship with inflammation begins.
Elevated mitochondrial ROS activates the NLRP3 inflammasome, a multi-protein complex inside immune cells that acts as a sensor for cellular danger. Once activated, the NLRP3 inflammasome triggers the release of pro-inflammatory cytokines, particularly interleukin-1β and interleukin-18. Research published in Nature Immunology found that the mitochondrial electron transport chain is directly necessary for NLRP3 activation, pointing to the centrality of mitochondrial function in this signaling pathway (Billingham et al., 2022).
A second pathway involves mitochondrial dysfunction and the release of mitochondrial DNA into the cytosol. When mitochondria are damaged and not cleared efficiently, fragments of their DNA can leak out. Because mitochondrial DNA carries molecular signatures resembling bacterial DNA (a remnant of evolutionary history), the immune system can recognize it as a danger signal and mount an inflammatory response. This mechanism, mediated in part by the cGAS-STING pathway, illustrates how impaired mitochondrial quality control contributes to inflammation that has no external pathogen to fight.
How Inflammation Damages Mitochondria in Return
Here is where the two-way relationship becomes particularly important. Chronic inflammation does not just result from mitochondrial dysfunction. It also feeds back and worsens it.
Pro-inflammatory cytokines can directly impair mitochondrial function, reducing the efficiency of the electron transport chain, lowering ATP output, and increasing ROS production further. This creates a self-reinforcing cycle: dysfunctional mitochondria generate more ROS, more ROS drives more inflammation, and more inflammation further impairs mitochondrial capacity. Reviews published in Signal Transduction and Targeted Therapy (Xu et al., 2025) describe this feedback as a core mechanism in how cellular oxidative stress becomes chronic rather than resolving.
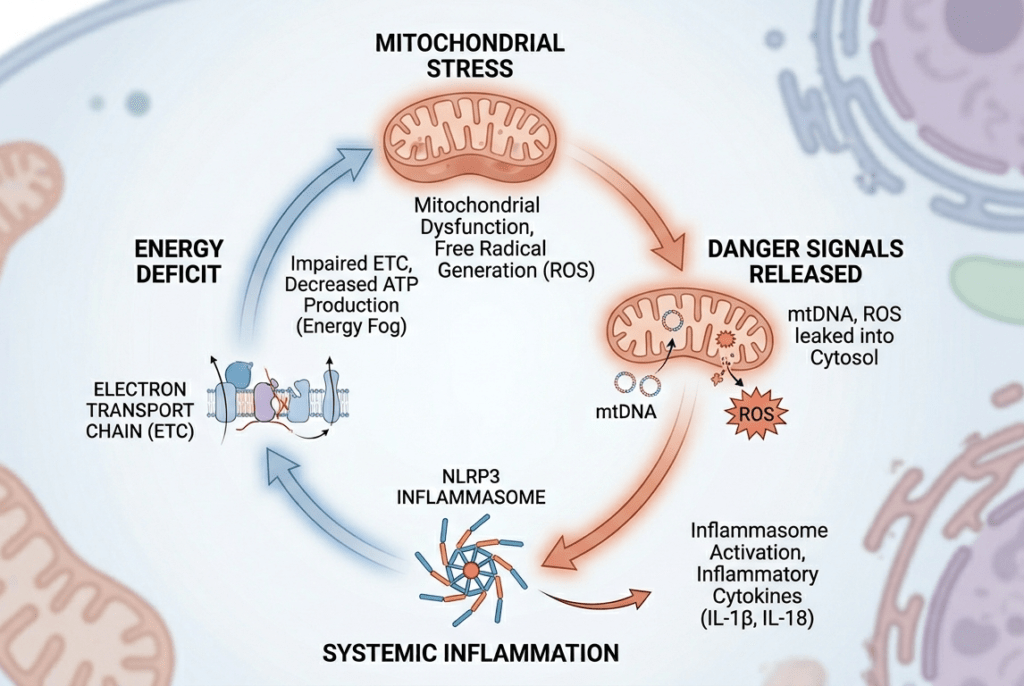
Inflammation also disrupts mitophagy, the cellular process by which damaged mitochondria are identified and removed. When mitophagy is impaired, dysfunctional mitochondria accumulate rather than being recycled. Research suggests that dysfunctional mitophagy can contribute to the transition from acute, protective inflammation to a more sustained, low-grade state (Gene Expression, 2023). A 2024 study published in Current Medicinal Chemistry found that cells with defective mitophagy lost the normal tolerance response to repeated inflammatory activation, with pro-inflammatory cytokine output continuing to rise rather than resolving, a pattern not seen in cells with intact mitophagy (Orekhov et al., 2024). This remains an active area of investigation, and the mechanisms involved are still being characterized in human contexts.
What the Research Shows
Human studies on the inflammation-mitochondria axis are still developing, but several lines of evidence are informative. A double-blind randomized clinical trial by Munguia et al. (2019) found that a high-flavonoid cocoa supplement in older adults was associated with reduced plasma oxidative stress markers and lower indicators of systemic inflammation, alongside improvements in mobility and quality of life. That study, which included (-)-epicatechin as a primary flavanol component, is available on the Mitozz peer-reviewed papers library.
Separately, cell model research has explored how (-)-epicatechin interacts with mitochondrial redox balance under conditions of oxidative stress. A study published in Oxidative Medicine and Cellular Longevity found that at relevant concentrations, epicatechin attenuated mitochondrial superoxide production in vascular endothelial cells under high-glucose conditions, and modulated mitochondrial complex activity, suggesting a role in supporting redox balance at the cellular level (Malin et al., 2020). These findings are preclinical and should not be interpreted as outcomes in human disease.
Lifestyle Factors That Support the Balance
The good news is that the mitochondria-inflammation axis is not fixed. Several well-supported lifestyle factors influence both sides of this relationship.
Regular physical activity is among the most studied. Exercise promotes mitochondrial biogenesis, improves mitochondrial efficiency, and has been shown to reduce markers of systemic inflammation across multiple population studies. The mechanism is partly mitochondrial: trained cells produce less ROS relative to their energy output and clear damaged mitochondria more efficiently.
Sleep plays an underappreciated role as well. Sleep deprivation is associated with increased inflammatory markers and impaired mitochondrial function. The relationship appears to be dose-dependent, with even modest improvements in sleep quality showing measurable effects on redox and immune markers in some observational studies.
Nutritional factors, particularly adequate intake of antioxidant-rich foods and avoidance of chronic caloric excess, support the cell’s capacity to buffer oxidative stress. Stress load and recovery practices also matter: sustained psychological stress activates inflammatory pathways and has been linked to mitochondrial changes in immune cells.
None of these factors works in isolation. They interact as a system, and supporting mitochondrial function means attending to all of them as a whole rather than optimizing any single variable.
Where Mitozz Fits In
Mitozz is a nutraceutical developed by FMG Health Sciences containing 98% pure (-)-epicatechin, a flavanol found naturally in cacao and studied in relation to mitochondrial function, redox balance, and cellular energy. The research on (-)-epicatechin and oxidative stress, while still developing in human clinical contexts, points to a compound that interacts meaningfully with the biological systems described in this article, particularly the mitochondrial redox pathways involved in inflammatory signaling.
Mitozz is designed to complement the lifestyle foundations described above, not replace them. If you are curious about how (-)-epicatechin fits into your daily routine, learn more at Mitozz product page.
The Bottom Line
Mitochondria and inflammation are not separate topics. They are deeply interconnected, each capable of influencing the other in ways that compound over time. Dysfunctional mitochondria generate the signals that drive persistent inflammation, and persistent inflammation in turn impairs the mitochondria responsible for cellular resilience and energy.
Supporting this system through movement, sleep, nutrition, and stress recovery is not a single intervention but an ongoing practice, one that operates at the level of your cells whether or not you are thinking about it.
Comparte este artículo del blog
Referencias
- Billingham, L. K., Revelo, M. P., & Bhatt, D. L. (2022). Mitochondrial electron transport chain is necessary for NLRP3 inflammasome activation. Nature Immunology, 23, 692–704.
- Kim, M. E., Lim, Y., & Lee, J. S. (2025). Mitochondrial dysfunction and metabolic reprogramming in chronic inflammatory diseases. Current Issues in Molecular Biology, 47(12), 1042.
- Malin, S. K., et al. (2020). (–)-Epicatechin modulates mitochondrial redox in vascular cell models of oxidative stress. Oxidative Medicine and Cellular Longevity.
- Orekhov, A. N., et al. (2024). Defective mitophagy impairs response to inflammatory activation of macrophage-like cells. Current Medicinal Chemistry.
- Munguia, L., et al. (2019). High flavonoid cocoa supplement ameliorates plasma oxidative stress and inflammation levels while improving mobility and quality of life in older subjects. The Journals of Gerontology Series A.Read on Mitozz
- Role of Mitochondria in the Chronification of Inflammation: Focus on Dysfunctional Mitophagy and Mitochondrial DNA Mutations. (2023). Gene Expression.
- Xu, X., Pang, Y., & Fan, X. (2025). Mitochondria in oxidative stress, inflammation and aging: From mechanisms to therapeutic advances. Signal Transduction and Targeted Therapy, 10, 190.
Comprender la salud mitocondrial es un proceso a largo plazo y por eso hemos creado la Mitozz . Se trata de un espacio gratuito para explorar la ciencia de la energía celular, aprender cómo los hábitos de vida favorecen a las mitocondrias y mantenerse informado a través de debates con expertos, contenidos educativos y sesiones de preguntas y respuestas en directo, a tu propio ritmo.
Únete a la Mitozz
Accede a debates entre miembros y expertos, contenidos formativos y sesiones de preguntas y respuestas en directo centrados en la energía celular y la salud mitocondrial.
Aviso médico: La información proporcionada en este artículo tiene fines exclusivamente educativos e informativos y no pretende constituir un consejo médico. No sustituye el diagnóstico, el tratamiento ni la orientación médicos profesionales. Consulte siempre a un profesional sanitario cualificado antes de realizar cambios en su dieta, rutina de ejercicio, prácticas de ayuno o uso de suplementos, especialmente si padece alguna afección médica, está embarazada o en periodo de lactancia, o está tomando medicamentos.
Descargo de responsabilidad de la FDA: Estas declaraciones no han sido evaluadas por la Administración de Alimentos y Medicamentos (FDA). No están destinadas a diagnosticar, tratar, curar ni prevenir ninguna enfermedad.






