Your cells each can contain anywhere from a few hundred to several thousand mitochondria depending on how energy-demanding they are, and collectively across your body, they number in the quadrillions. From the moment you wake up, they convert nutrients into ATP, the molecule that powers virtually every biological process in your body. By the time you fall asleep, many of those mitochondria are carrying wear from the day: oxidative byproducts, structural changes, and accumulated molecular stress.
Sleep, it turns out, is when the cell settles that debt.
Sleep Is a Biological Maintenance Window
For decades, sleep was largely studied through the lens of the brain, memory consolidation, hormonal rhythms, and immune activity. That framing was accurate but incomplete. A growing body of research now points to sleep as a critical period of mitochondrial quality control, when cells actively clear damaged components, produce new mitochondria, and restore the energetic capacity needed for the next day.
Research in sleep neurobiology suggests that the cycling of sleep and wakefulness reshapes neuronal activity, gene expression, and cellular metabolism, with mitochondria playing a central mediating role. This isn’t background maintenance in the conventional sense. It’s a coordinated biological process that depends on sleep to run properly.
What Happens Inside the Cell While You Rest
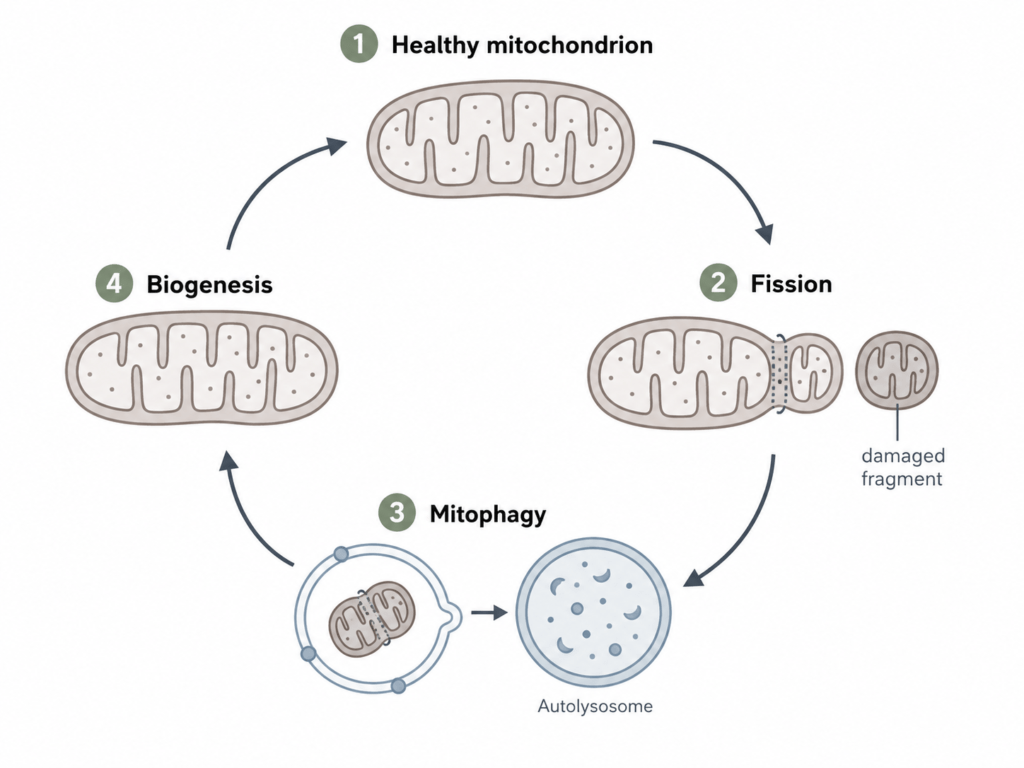
Two interconnected processes are central to mitochondrial renewal during sleep: mitophagy and mitochondrial biogenesis.
Through mitophagy, a selective form of cellular cleanup, cells remove and break down damaged or dysfunctional mitochondria, preventing the buildup of oxidative stress and inflammation. A significant portion of mitochondrial mass is replaced on a daily basis through this quality control process. Think of it as the cell’s nightly audit: anything that isn’t functioning efficiently gets flagged and cleared.
Biogenesis is the other side of that process. While mitophagy handles removal, biogenesis generates new mitochondria by activating specific gene expression programs. Research suggests that during recovery sleep, cells upregulate mitochondrial gene expression to prime the production of new organelles, restoring bioenergetic capacity after periods of high activity. The cell isn’t just resting, it’s rebuilding.
Why does this matter for how you feel?
Because your capacity to sustain energy throughout the day depends in part on how well this renewal cycle runs. A well-maintained mitochondrial network produces ATP more efficiently, handles metabolic flexibility more effectively, and generates fewer harmful byproducts. The maintenance window sleep provides is a meaningful contributor to that capacity.
When Sleep Is Disrupted, So Is Mitochondrial Function
Research suggests a bidirectional relationship between mitochondrial dysfunction and sleep quality, with sleep deprivation impairing mitochondrial biogenesis and function, while mitochondrial disorders are often associated with sleep disturbances. The relationship runs in both directions, which makes consistent sleep a meaningful lever for cellular health. If you want to understand what mitochondrial dysfunction can feel like at a lived level, this breakdown of common patterns is a useful reference.
One of the more studied consequences of poor sleep is its effect on oxidative stress and redox balance. Prolonged wakefulness leads to increased levels of reactive oxygen species and a reduction in antioxidant defenses. Sleep deprivation may also impair mitochondrial function and reduce ATP production. Over time, if the cell can’t clear this oxidative load, the mitochondria that remain are working under more strain.
Even a single night of sleep deprivation in human subjects has been observed to alter systemic redox metabolites, including plasma antioxidant levels such as glutathione. This doesn’t mean one bad night undoes your cellular health, but it does suggest the mitochondrial system is sensitive to sleep patterns in ways that show up quickly in measurable markers.
Signs Your Cellular Energy mayo Be Under Strain
There’s no single symptom that maps cleanly to mitochondrial stress. But some common experiences, especially when they occur together and persist, may reflect the kind of metabolic strain that builds when sleep-dependent maintenance is chronically disrupted.
Persistentfatigue that isn’t explained by physical exertion, difficulty recovering from exercise, cognitive sluggishness despite adequate caffeine, and a general sense of low reserve are all patterns that researchers studying cellular energy metabolism find relevant. These aren’t diagnostic markers, and many factors contribute to how energized or depleted someone feels. But they’re worth noting as signals that your systems may not be running a full recovery cycle overnight. Our article on how to repair and maintain mitochondrial health naturally covers the broader landscape of habits that support this process.
How to Support Mitochondrial Recovery Through Sleep
The evidence around sleep quality and mitochondrial health points toward a few consistent behavioral themes. None of these are novel, but the cellular mechanism behind them gives them more weight than standard sleep hygiene advice usually conveys.
Maintaining consistent sleep and wake times supports the circadian regulation of mitochondrial quality control. Mitochondria’s optimal functioning is closely linked to the body’s circadian clock, and irregular schedules may disrupt the timing of cellular renewal processes, even when total sleep time is adequate.
Limiting bright light in the evening, avoiding stimulants close to bedtime, and moderating alcohol intake all support the deeper, slower sleep stages where restorative biological processes are most active. Therapeutic strategies targeting sleep disturbances may support resilience against oxidative stress and overall cognitive function.
Metabolism and nutrition also play a role. Mediterranean-style diets that prioritize plants and minimally processed foods have been associated with mitochondrial health and reduced oxidative stress, which complements what adequate sleep is doing at the cellular level.
Movement, hydration, and stress management round out the picture. One thing the research consistently reinforces is that long-term consistency matters more than short-term intensity when it comes to supporting mitochondrial function.
Sleep works alongside these foundations, not instead of them.

Where Mitozz Fits In
Mitozz is a nutraceutical developed by FMG Health Sciences containing 98% pure (-)-epicatechin, a flavanol found in cacao and green tea that has been studied for its effects on mitochondrial function and cellular energy capacity.
Research in this area has examined (-)-epicatechin in relation to mitochondrial biogenesis and oxidative stress. Studies in animal models have explored the capacity of (-)-epicatechin to reduce aging-induced oxidative stress and support indicators of mitochondrial biogenesis across multiple tissue types, including brain, heart, kidney, and skeletal muscle. You can read that study in full on its Mitozz research page. Separately, reviews of natural compounds including (-)-epicatechin have summarized evidence suggesting improvements in mitochondrial biogenesis and electron transport chain function across a variety of experimental systems.
This research is ongoing, and most of it has been conducted in preclinical or early-stage human settings. Mitozz is not a treatment for sleep disorders or mitochondrial disease. It’s designed as a complement to the lifestyle fundamentals that support cellular energy, among them consistent, quality sleep.
For a deeper look at how (-)-epicatechin and mitochondrial support relate, this overview of the science behind Mitozz is a good starting point. We recommend talking with a healthcare provider about what makes sense for your situation.
The Bigger Picture
Sleep is one of the most underappreciated tools for long-term cellular health. Not because it’s passive, but because it’s when some of the most active biological maintenance in your body happens to take place. The mitochondria in your cells depend on that window to clear damage, rebuild capacity, and set up the energy systems that carry you through the next day.
Understanding the cellular reasons behind a good night’s sleep doesn’t change the basic advice, but it does reframe it. Rest isn’t recovery from activity, it’s part of the biological process that makes sustained activity possible. That’s a distinction worth holding onto.
Referencias
- Mauri, S., Favaro, M., Bernardo, G., Mazzotta, G. M., & Ziviani, E. (2022). Mitochondrial autophagy in the sleeping brain. Frontiers in Cell and Developmental Biology, 10, 956394.
- Sarnataro, R., Velasco, C. D., Monaco, N., Kempf, A., & Miesenböck, G. (2025). Mitochondrial origins of the pressure to sleep. Nature. Read on Mitozz
- Sarnataro, R. (2025). Neurobiology of mitochondrial dynamics in sleep. The Journal of Physiology.
- Frontiers in Aging. (2025). Unraveling the interplay between sleep, redox metabolism, and aging: implications for brain health and longevity. Frontiers in Aging, 6, 1605070.
- Cellular and Molecular Neurobiology. (2024). Sleep and oxidative stress: current perspectives on the role of NRF2. Cellular and Molecular Neurobiology.
- Institute for Functional Medicine. (n.d.). Sleep deprivation: mitochondrial impact and therapeutic strategies. Institute for Functional Medicine.
- Moreno-Ulloa, A., Nogueira, L., Rodriguez, A., et al. (2015). Recovery of indicators of mitochondrial biogenesis, oxidative stress, and aging with (-)-epicatechin in senile mice. The Journals of Gerontology: Series A, 70(11), 1370–1378. Read on Mitozz
- Gonzalez Garza, M. T., & Cruz-Vega, D. E. (2020). Effects of (-)-epicatechin on mitochondria. Nutrition Reviews, 79(1), 25–41.
- Varela, C. E., et al. (2021). Regulation of cytochrome c oxidase by natural compounds resveratrol, (-)-epicatechin, and betaine. International Journal of Molecular Sciences, 22(12), 6606.







